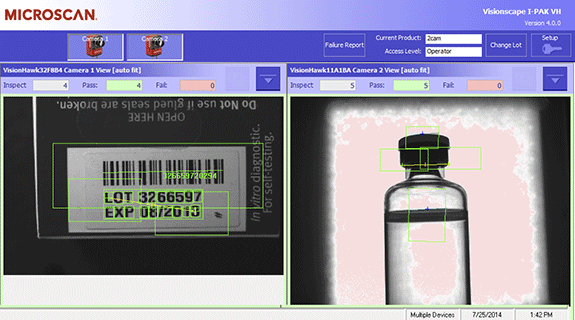
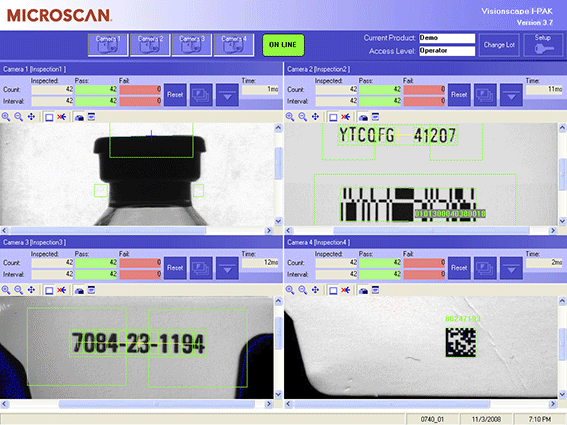
- Multi-camera visualization on a single screen
- Troubleshooting tools to identify problems and reduce downtime, including image failure queue with time and date stamps and detailed per-camera failure reports
- Controlled system access based on user ID and password
- Support for color vision tools
- Verifies symbol readability immediately after printing
- Text reading and verification with OCR and OCV
- Optional 21 CFR Part 11 compliance for FDA-regulated industries
Visionscape® I-PAK® Software is a proven inspection solution chosen by major manufacturers worldwide. Combining powerful machine vision tools with a simple user interface, I-PAK meets demanding industry requirements for reliable and accurate inspection of labels and product assemblies. Typical applications include barcode and OCR, print quality verification, color matching and inspection, defect detection, date/lot code verification, component ID verification, and a variety of other package, label, or product inspections.

I-PAK® VH: I-PAK user interface and support for up to four Vision HAWK Smart Cameras in standard or C-Mount models with fully integrated autofocus and color functionality.
Powerful Capabilities: I-PAK’s full suite of machine vision tools offers manufacturers quick and easy system setup for high-mix production lines. Supervisors can save specific settings for numerous inspection jobs, change lot numbers without exiting the runtime screen, scale manufacturing processes, and minimize downtime with diagnostic capabilities that quickly pinpoint failures for each inspection point.
Fail-Safe Identification: I-PAK identifies incorrect characters while tolerating normal print variation. OCV algorithms adapt to changes in preprinted or overprinted symbols to reduce false rejects.
FDA Compliance: I-PAK enables manufacturers to comply with the FDA’s current Good Manufacturing Practices. It meets the requirements of 21 CFR Part 11 through optional user access control features and audit trail capabilities.





Reviews
There are no reviews yet.